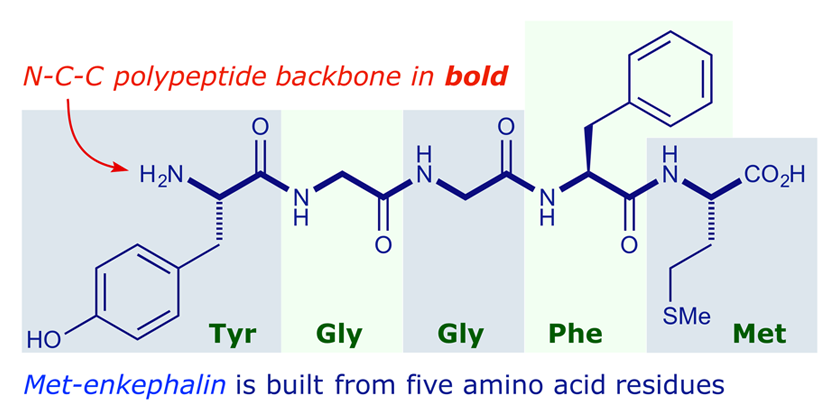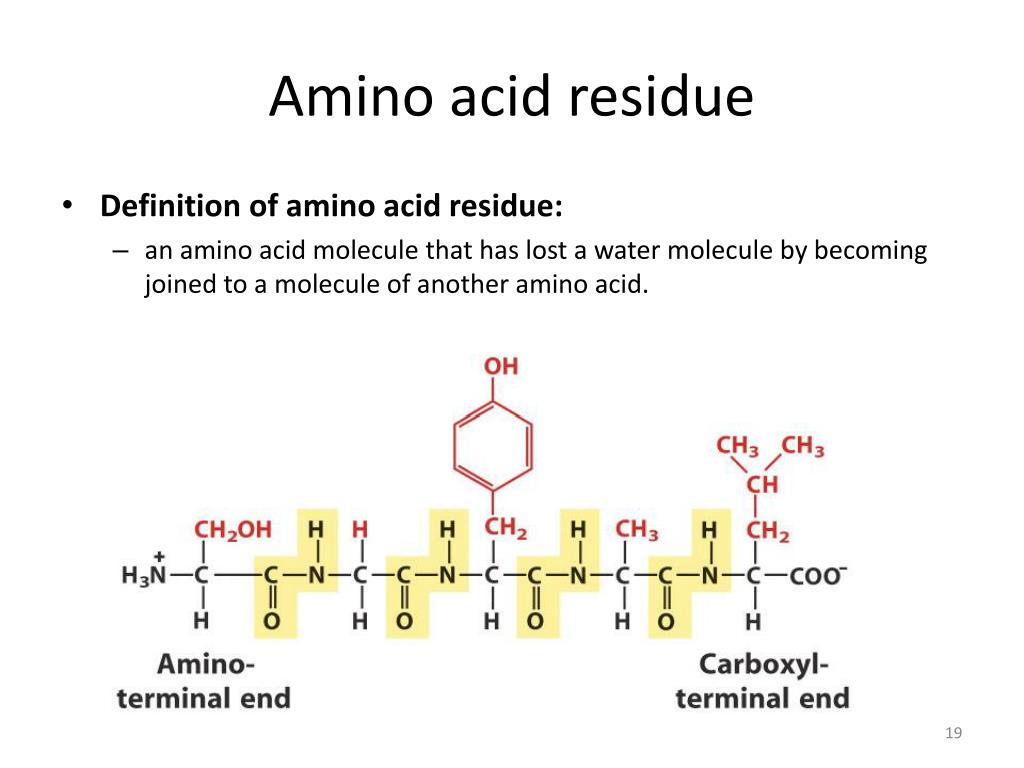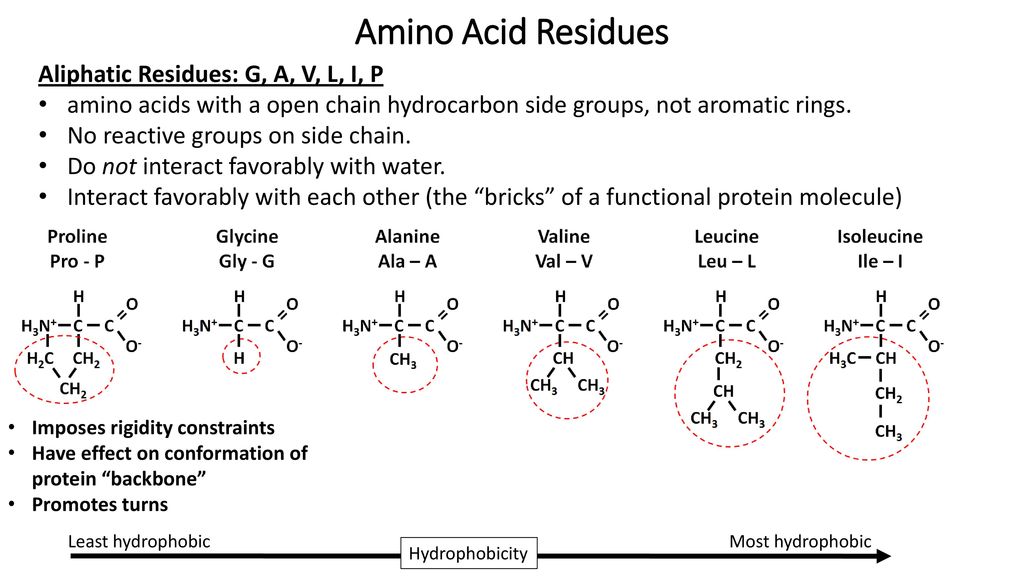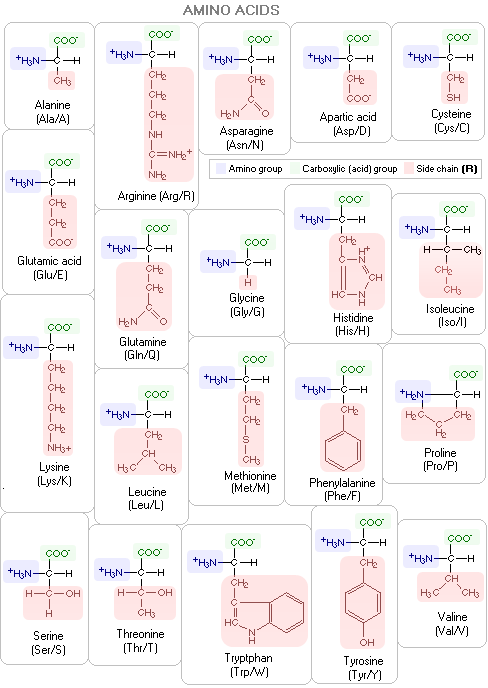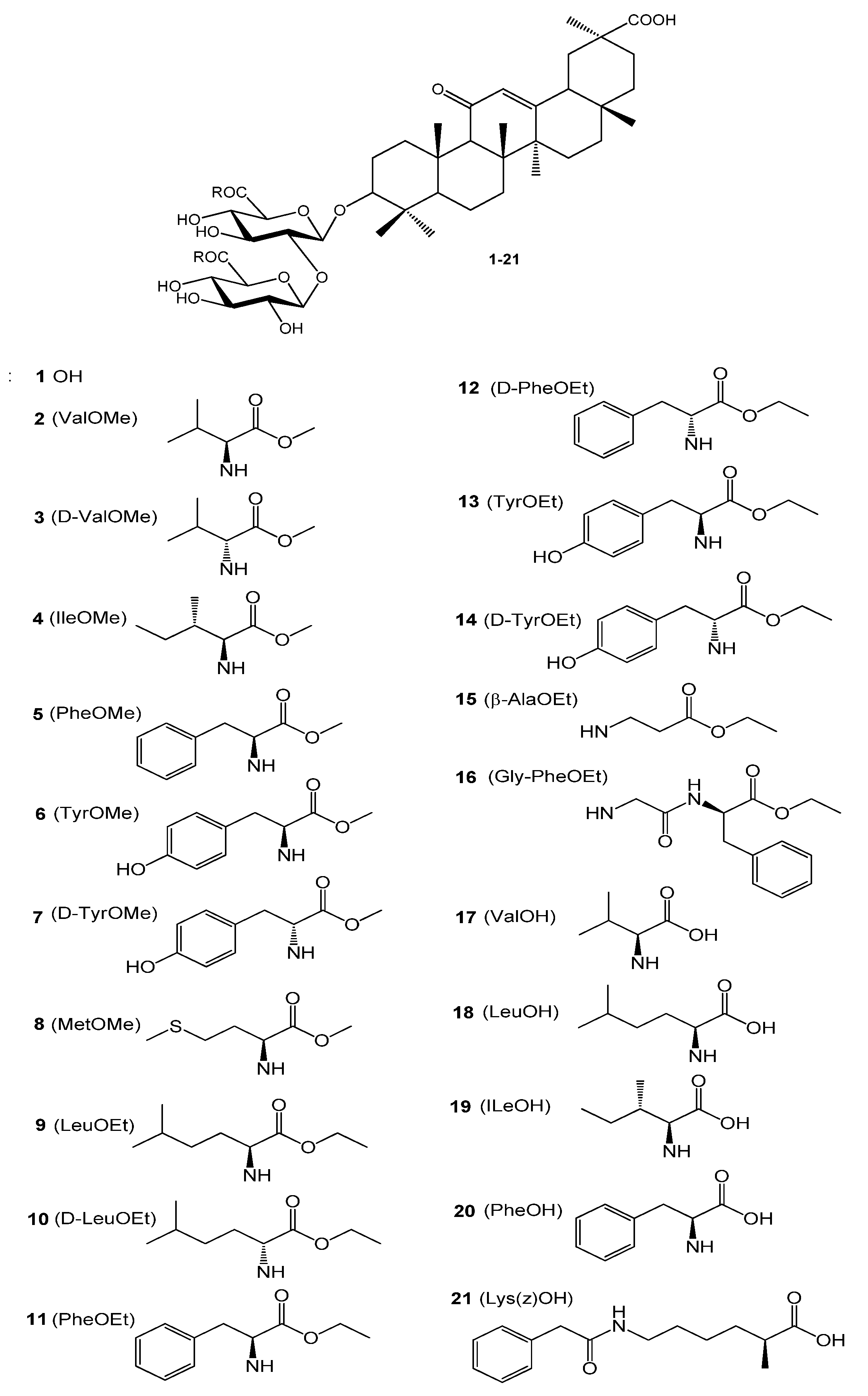
IJMS | Free Full-Text | Glycyrrhizic Acid Derivatives Bearing Amino Acid Residues in the Carbohydrate Part as Dengue Virus E Protein Inhibitors: Synthesis and Antiviral Activity

Thermodynamic Scale of β-Amino Acid Residue Propensities for an α-Helix-like Conformation | Journal of the American Chemical Society

Identification of Amino Acid Residues Involved in the Inactivation of Cytochrome P450 2B1 by Two Acetylenic Compounds: The Role of Three Residues in Nonsubstrate Recognition Sites | Journal of Pharmacology and Experimental

Endogenous l- to d-amino acid residue isomerization modulates selectivity between distinct neuropeptide receptor family members | PNAS